(b) Describe what you would observe when aqueous silver nitrate is added to separate aqueous solutions of Potassium Fluoride and Potassium Bromide. (ii) Explain why Bromine does not react with aqueous Chloride ions. Write an equation for the reaction occurring. (a) (i) Describe what you would observe when an aqueous solution of Bromine is added to an aqueous solution containing Iodide ions. 
Suggest Corrections 7 Similar questions Q. As we go from F 2 to I 2 th energy released in the propogating steps decreases which proves the reactivity order. Halogens become less reactive as you go down group 7 in the periodic table, because the outer electron shell gets further away from the attraction of the. In halogenation of alkanes, the mechansim is radical mechanism. 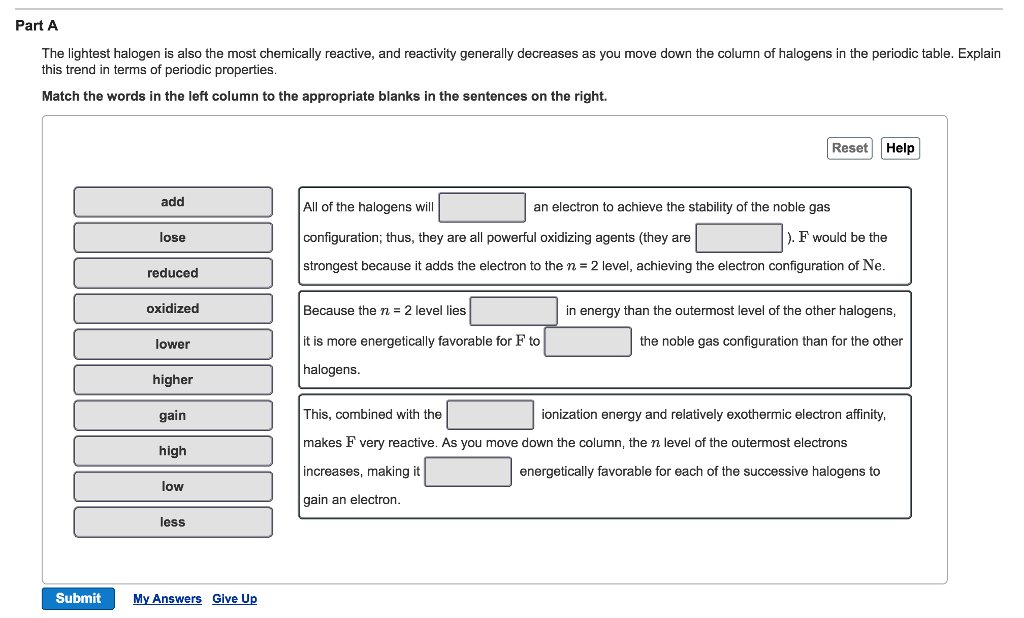
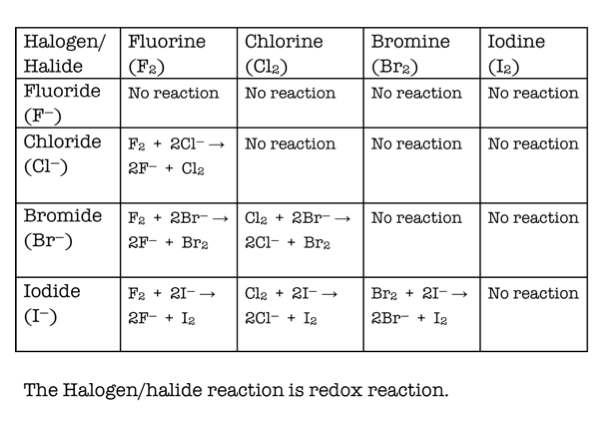
So there will be no reaction in the I 2/KCl mixture or the I 2/KBr mixtureīoth tubes takes the colour of the Iodine - brown if concentrated enough, yellow if very dilute. In halogenation of alkanes, reactivity of halogens decrease with decrease in the electronegativity. We should expect this because Iodine is larger than Br and Cl, so not capable of taking electrons from either ions. When KCl and KBr have Iodine water added there is no colour change in either tube. The tube takes the colour of the Iodine - brown if concentrated enough, yellow if very dilute Reactivity decreases as you move down the column. The last three tubes: Adding Iodine water to KI is a waste of time. Fluorine is the most reactive and combines with most elements from around the periodic table. This means that in the Br 2/KI mixture Bromine will be converted to Bromide ions while and Iodine will be made from Iodide ions.īut there will be no reaction in the Br 2/KCl mixtureīr 2(aq) + 2I - (aq) → 2Br - (aq) + I 2(aq) So, Bromine is capable of taking electrons from Br - ions but not from Cl - ions. We should expect this because Bromine is smaller than I but not Cl. When KCl and KI have Bromine water added there is a colour change in one tube only. The next three tubes Adding Bromine water to KBr is a waste of time. Describe the redox reactions of the Group 7 elements with other halide ions. (note that Group 1 halide solutions have no colour ) Explain the trend in reactivity for the Group 7 elements. Observations: Slightly green/yellow solution goes orange. They are unusual in that they are coloured and diatomic. Observations: Slightly green/yellow solution goes brownĬl 2(aq) + 2Br - (aq) → 2Cl - (aq) + Br 2(aq) The halogens, group 7, are an important group of elements both chemically and socially. This means that Chlorine will be converted to Chloride ions while Bromine and Iodine will be made from Bromide and Iodide ions.Ĭl 2(aq) + 2I - (aq) → 2Cl - (aq) + I 2(aq) We should expect this because Chlorine is smaller than Br or I, so capable of taking electrons from Br - or I - ions. The next two tubes: When KBr and KI are added to Chlorine water there is a colour change in both tubes. The tube stays colourless (or at least very pale yellow) The first tube: Adding Chlorine water to KCl is a waste of time. This is an experiment you may well do, or have already done.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
